What are the Latest Trends in Pharma Procurement Strategy?
- Procurement strategies built for resilience outlast those built for cost efficiency.
- Multi-hub sourcing ensures one regional disruption never becomes a global shortage.
- CDMOs with constrained capacity choose their strategic partners over transactional buyers every time.
April 01, 2026 | Pharma and Life Sciences 8 minutes read
While most industries tolerate procurement inefficiency, pharma cannot. A single sourcing failure can delay a drug launch, violate compliance mandates, or worse, pull a product from the market. The good news is that staying ahead of the latest trends in pharma procurement strategy will be a competitive differentiator.
Pharmaceutical procurement strategies have evolved faster in the last three years than in the previous decade.
This article breaks down what's actually changing in pharma procurement, why it matters at the leadership level, and where your focus should be if you want to stay ahead.
Scale Pharma Operations with an Integrated Supply Chain
Align global sourcing and logistics into a single, high-performance engine
The Role of Pharma Procurement in Modern Supply Chains
Pharma procurement isn't just about buying raw materials and active pharmaceutical ingredients (APIs) at the lowest cost. It's the connective tissue between R&D timelines, manufacturing schedules, regulatory compliance, and patient outcomes, operating across a multi-tiered, globally distributed supply chain where any weak link can trigger cascading consequences.
From cold-chain logistics and serialization requirements to single-source supplier dependencies and geopolitical risk, the pharma supply chain operates under a level of complexity that few other industries face.
Procurement sits at the center of it all, managing relationships, contracts, risk exposure, and spend visibility simultaneously.
But complexity alone isn't the issue. Strategy is.
The pharmaceutical industry's procurement landscape demands a forward-looking strategy over a reactive one. It must anticipate supply risk, build optionality into the supplier base, integrate compliance requirements into sourcing decisions from day one, and align procurement goals with broader business objectives.
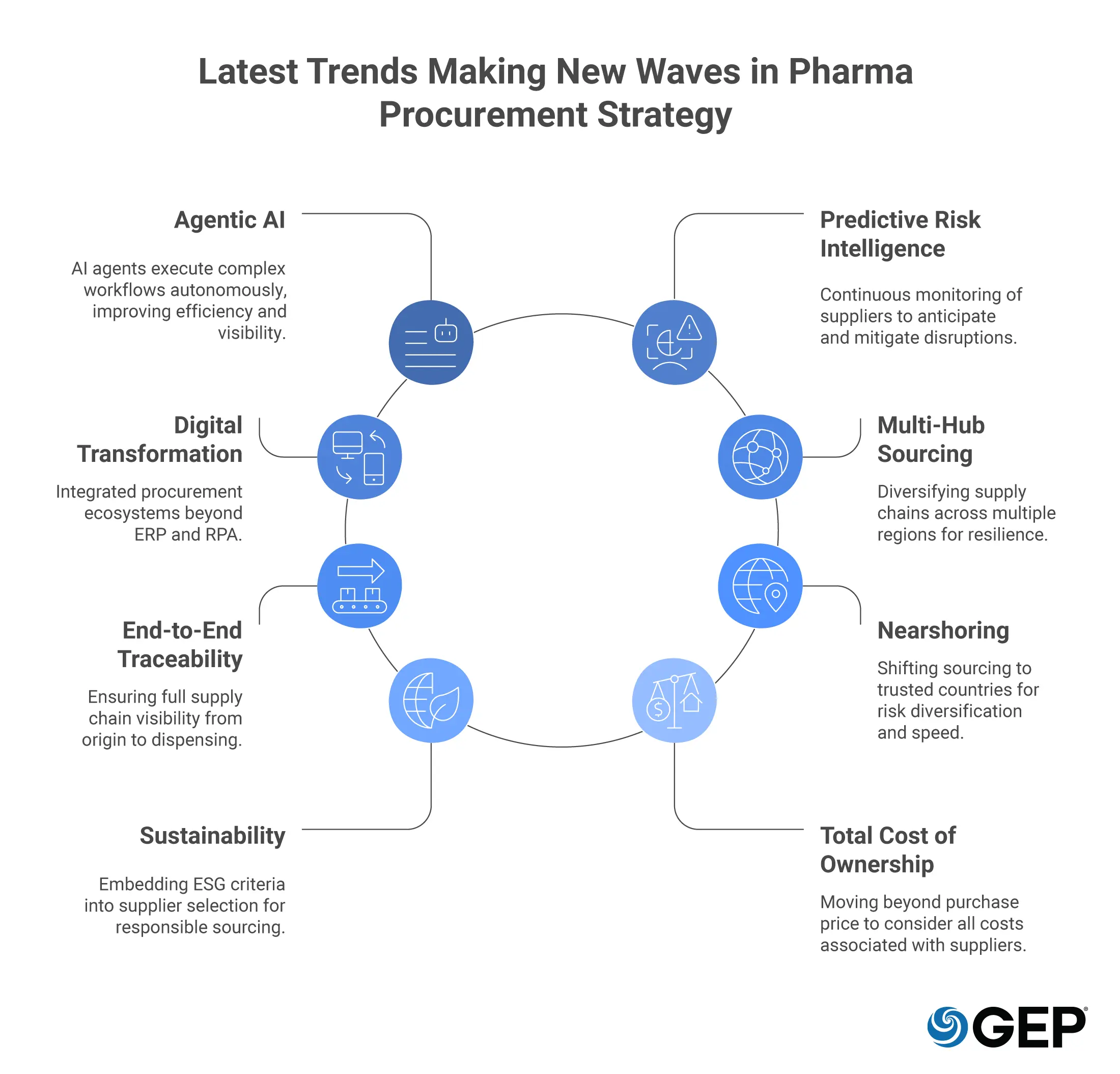
9 Trends in Pharma Procurement Strategy Shaping Resilient Supply Chains
The pharma procurement landscape is shifting on multiple fronts at once, from how AI executes workflows to how geopolitical legislation is forcing entire supply chains to be rebuilt.
All of the latest trends in the pharmaceutical industry require leadership-level attention for firms that want to have structural advantages that can't be replicated in a hurry.
1. Agentic AI Is Redefining What Procurement Can Do
The biggest shift happening right now isn't automation. It's autonomy.
AI agents in procurement can now execute multi-step workflows without human intervention. They can do everything independently, from analyzing supplier performance data and flagging risk signals to initiating RFQs, evaluating bids against predefined criteria, and escalating exceptions to the right stakeholders.
What separates agentic AI from traditional automation is that it doesn't just complete tasks; it reasons through them.
For pharma procurement leaders, deploying AI agents across source-to-pay is seeing faster cycle times, fewer contract compliance gaps, and significantly better spend visibility.
2. Supplier Risk Intelligence Is Becoming Predictive
Reactive risk management, where you respond to a supplier failure after it hits your supply chain, is no longer acceptable in pharma. The new standard is predictive risk intelligence.
Your procurement platform must continuously monitor suppliers across financial health, geopolitical exposure, regulatory status, production capacity, and ESG compliance, and bring early warning signals to leaders before they become disruptions.
Pharma procurement trends are moving decisively toward real-time supplier intelligence platforms that integrate external data feeds, including news, regulatory filings, and financial indicators, with internal supplier performance data to give your team a live risk picture at all times.
3. Digital Transformation in Procurement Beyond ERP and RPA
Digital transformation in procurement used to mean implementing an ERP and calling it a day. That era is over.
Leading pharma organizations are now building integrated procurement ecosystems: AI-powered spend analytics, intelligent contract lifecycle management, supplier collaboration portals, and demand-sensing tools that connect procurement decisions to real-time manufacturing and distribution signals.
What makes this transformation truly powerful is the data layer it creates.
When every procurement decision and execution is captured and analyzed, you start to see patterns that were previously invisible. Pricing anomalies, maverick spend, contract leakage, and supplier consolidation opportunities are critical insights that make your procurement function progressively smarter and resilient.
4. Multi-Hub Sourcing Is Replacing Single-Region Supply Dependency
Decades of efficiency-driven globalization left pharma supply chains dangerously concentrated. The lesson learned is that having all eggs in one basket, whether in one country or one facility, is dangerous.
Companies are now striving to build a network of regional hubs through manufacturing resilience hubs. Facilities are designed with excess capacity and flexible production lines that can step in if another site goes down or demand surges.
For procurement strategy, multi-hub sourcing deliberately qualifies suppliers across multiple geographies, building redundancy into critical categories before disruption forces the issue, and treats geographic diversification as a standard criterion in supplier selection.
The shift toward diversified, regionalized, and technologically advanced supply chains is becoming crucial for ensuring uninterrupted access to medications globally.
5. End-to-End Traceability Is Becoming a Procurement Requirement
Regulatory bodies across major markets are tightening serialization and traceability mandates, with full supply chain visibility from raw material origin through to the dispensing point.
Traceability is now a sourcing criterion. Suppliers that can't provide robust documentation, site-level transparency, change-control communication, and material origin data are becoming a liability regardless of their price point.
Many investigated drug shortages stem from weaknesses in external supply chain control rather than manufacturing failures. Procurement teams that embed traceability requirements into supplier qualification processes can significantly reduce regulatory exposure.
6. Nearshoring is Reshaping Sourcing
The broader geographic diversification trend is now accelerating across the industry. Companies are shifting toward trusted countries with strong regulatory frameworks and talent pools, enabling biopharma sponsors to diversify risk, access specialized resources, and increase time-to-market while ensuring a robust, reliable supply chain.
Rethink category strategy at a geographic level, where suppliers in specific regions are prequalified as viable alternatives before a disruption forces a decision.
7. Sustainability and ESG Are Now Sourcing Criteria
Pharma companies are under increasing pressure from regulators, investors, and payers to demonstrate responsible sourcing.
Your procurement strategy needs to embed ESG criteria, including carbon footprint, ethical labor practices, water usage, and waste management, directly into supplier qualification and selection processes.
Suppliers with poor ESG performance carry hidden financial and regulatory risks. The pharmaceutical procurement strategies that will hold up over the next decade are those that treat sustainability not as a separate initiative but as a core dimension of supplier assessment and total cost of ownership calculations.
Also Read - Decoding ESG Reporting: A Comprehensive Guide
8. Total Cost of Ownership Is Replacing Purchase Price as the Primary Metric
The historical mandate of pharmaceutical procurement, to secure raw materials at the lowest possible unit cost, has been rendered obsolete by the increasing complexity of global supply chains.
The modern procurement function operates as a strategic orchestrator of value, risk, and innovation. Moving away from Purchase Price Variance and replacing it with Total Cost of Ownership (TCO) models that account for quality failures, supply disruptions, regulatory non-compliance costs, and the hidden costs of single-source dependency.
For procurement leaders, this shift has real organizational implications. It requires procurement to work more closely with all functions to build cost models that reflect true risk-adjusted value.
It also requires supplier segmentation strategies that allocate your relationship investment where it actually moves the needle, rather than spreading attention evenly across a vendor base of wildly varying strategic importance.
9. Strategic Supplier Partnerships Over Transactional Relationships
The days of treating API manufacturers and Contract Development and Manufacturing Organizations (CDMOs) as interchangeable vendors are over. Capacity is constrained, lead times are long, and the most capable suppliers have choices about who they work with.
Pharma procurement leaders who have invested in genuine strategic partnerships, built around joint planning, shared risk, and co-investment in capacity, are getting priority access and better terms.
In 2026, the bottlenecks will be tech transfer readiness, quality culture, analytics capability, and on-time deviation closure. Buyers will increasingly evaluate CDMOs like long-term partners, not vendors.
This trend is accelerating as more pharma companies expand biologics and cell and gene therapy pipelines, which require highly specialized manufacturing capabilities that can't be sourced on short notice.
Your procurement strategy needs to identify which supplier relationships deserve deep investment and build structured governance models around them.
The Agentic AI Playbook for Procurement Pros
Your roadmap to move from pilots to production, with AI that adapts, learns, and delivers
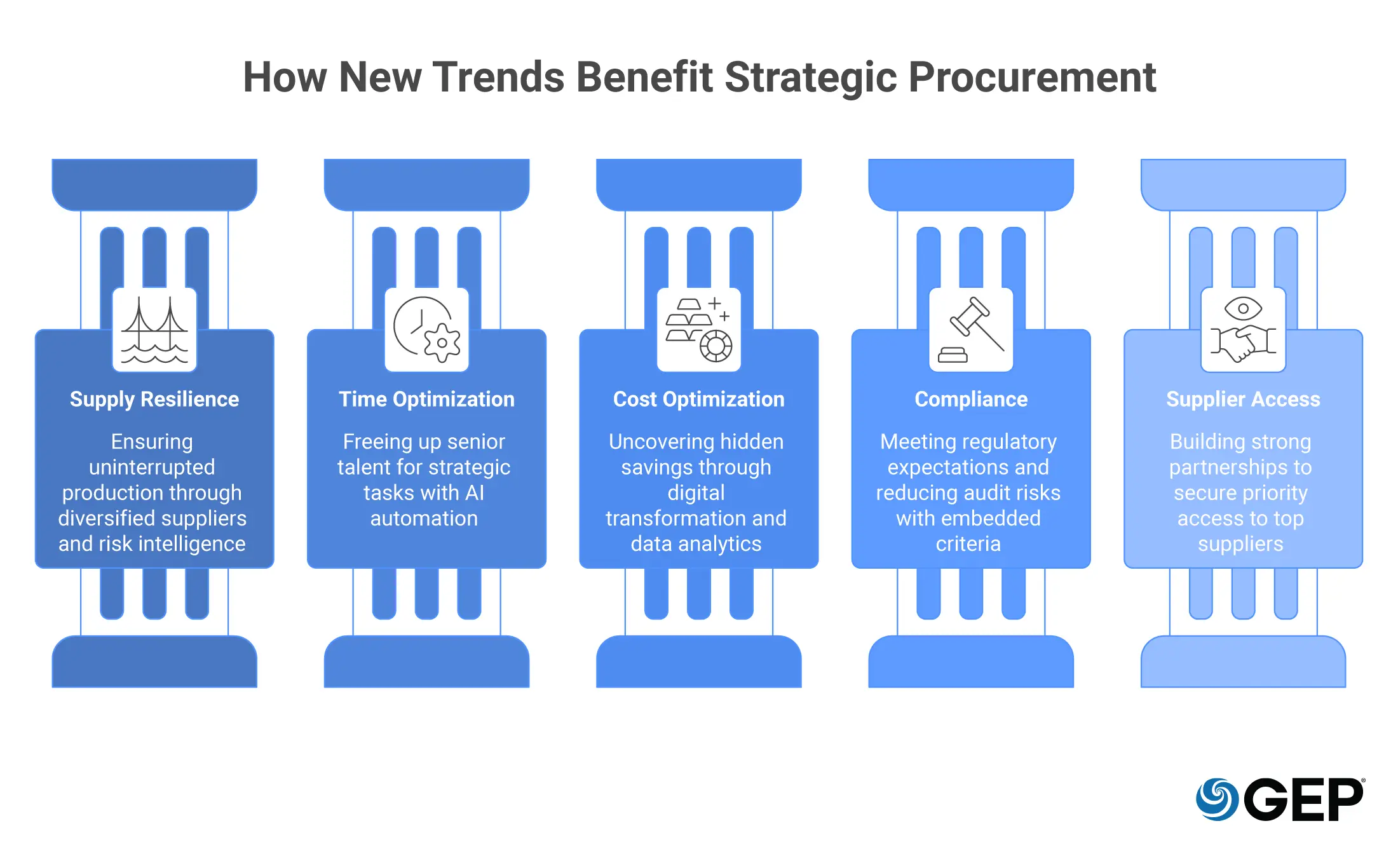
Top Five Benefits of Adopting Strategic Pharma Procurement Trends
Each capability you build compounds the value of the next one, and together they create a procurement function difficult for competitors to replicate.
1. Greater Supply Resilience
Every supply disruption that blindsides your organization is a failure of procurement strategy. Predictive risk intelligence and diversified supplier strategies are key.
When you have two or three pre-qualified, audit-ready suppliers for your most critical materials, and one of them goes down, you can switch without halting production, delaying a launch, or scrambling to qualify a backup under pressure.
2. Give Your Best People Their Time Back
Senior procurement talent shouldn't be chasing down purchase order status updates or manually consolidating supplier performance reports. AI agents and real-time data can handle that now. They can focus on the decisions that actually require judgment, like negotiating complex supplier agreements, evaluating strategic partnerships, and anticipating risks before they materialize.
3. Cost Optimization Beyond Price
Digital transformation surfaces contract leakage, maverick spend, unconsolidated categories, and supplier relationships that cost far more to maintain than they deliver in value. The organizations that see this clearly are unlocking cost reduction that no amount of negotiation pressure could have found.
4. Get Ahead of Regulators with Stronger Compliance Posture
Regulatory expectations around sourcing transparency, ESG performance, and supply chain documentation are tightening across every major market. Embedding these criteria directly into your sourcing and supplier qualification processes acts as a risk shield for procurement.
5. A Competitive Supplier Access Advantage
The best CDMOs, API manufacturers, and specialty suppliers are not sitting idle, waiting for your RFQ. Capacity is constrained, and the most capable partners are choosing who they work with based on relationship depth, not just contract value.
The procurement teams that have invested in strategic partnerships, such as joint planning, shared forecasting, and mutual accountability, will be given priority when capacity is tight. These relationships are built over time, and they pay off exactly when they matter most.
Staying Competitive with Evolving Pharma Procurement Trends
The next few years will separate pharma companies with intelligent procurement functions. AI agents will take on increasingly complex procurement workflows and supplier ecosystems, consolidating them around strategic partnerships. Regulatory compliance timelines for evolving mandates and drug-pricing reforms will intensify in every major market.
As a procurement leader, you must treat procurement as a strategic intelligence layer and invest in the right technical capabilities and data architecture. Restructuring your supplier relationships around strategic value and rethinking your sourcing geography will help you meet your business demands in a volatile market.
If you're ready to build a procurement function that performs at that level, exploring a structured procurement strategy framework is the right starting point, one that aligns your sourcing decisions with long-term business objectives and equips your organization to lead through whatever disruption comes next.
FAQs
Over-concentrated supplier bases leave you exposed the moment a region faces regulatory, logistical, or geopolitical pressure. Fragmented data, rising compliance demands, and most importantly, the lack of skilled talent required to navigate all of this is scarce. To add to this, the rising cost pressure from payers and the margin squeeze that comes with managing increasingly complex biologics and specialty drug pipelines is another critical challenge.
Sustainability is now becoming a sourcing decision. Investors, regulators, and institutional buyers are now asking hard questions about where materials come from, under what conditions, and at what environmental cost. And they want documentation to prove it. ESG criteria must be reflected within your supplier qualification process, contract terms, and ongoing performance reviews. Suppliers who can't meet these standards become a financial and regulatory liability.